Hello!
We do a lot of spinning and washing of cells on our Hamilton and I’m trying to figure out the best way to aspirate the supernatant after centrifugation. Ideally I could use liquid following but when I do that, the tip often hits the pellet (there is some variability in the volumes so I am overaspirating to make sure the residual volume is the same in all the wells). My workaround has been to create a plate with dead volume about the size of the cell pellet. This works great and I can do liquid following, but now I want to be able to transfer the cells but I can’t get below the dead volume. Although the dead volume works, I am hoping someone has found a better way… Thanks!
you can create another labware and use the 2nd labware for cell resuspension.
slow aspiration would help, also use fixed height.
Then move up and to the same again in the same well to remove as much of liquid that might have collected in those 2-3 seconds.
Thank you for the responses. @Pascal-the slow aspiration makes sense and I will test that.
@chips-a-hoai For the 2nd labware, is it possible to change the labware mid-protocol? If so, can you give me a brief how-to?
You can just add another labware on top of your existing labware on the deck editor. This will create automatically create another sequence which you can then use.
I’d suggest using Virtual Labware Library to adjust the pipetting heights here.
If you know the height which is the max pipette height for your plate not to disturb the pellet, you could virtually ‘move’ the plate by this height before the washing steps (so the tips don’t go to the very bottom). You’d just need to virtually move it back after the pipetting step.
This option does not require changing the labware definition or adding any additional labware to the deck ![]()
Thank you Chips and Magda! I’ll go ahead and try the adding labware on top first since that will be the easiest. The virtual labware sounds like a good option too if that doesn’t work out!
I used to use this library but don’t have it on my current machines - can you advise where one can find and download this?
@EricSindelar_Hamilton has all libraries shared for LabAutomation, you need to download VirtualLabware_V2 to make this possible.
Yes, this virtual labware library has been posted to the Hamilton Libraries shared folder.
You can also set a parameter in the container definition that would prevent the system from ever going past a specified height.
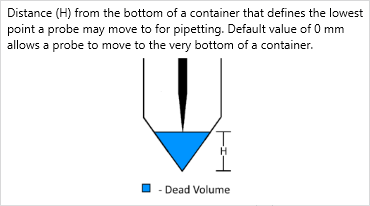
For VENUS five and prior, it’s referred to as Maximum pipetting height:
![]()
For VENUS six and above, it has been renamed to Dead volume height:
See below for more details on this parameter:
Maybe the BluePlate by BlueCatBio is an option - cells are deposited in cellsafe. It is designed to be used in the BlueWasher - but will also work in a ALH.
Hi Erik,
I’m developing a Red Blood Cell wash protocol that we’re running on a Hamilton STAR 4-channel system using a 24-well plate and integrated centrifuge.
We start with 50, 100, 150, or 200 µL sample, then add PBS to a total volume of 1000 µL. After spinning, I aspirate the supernatant with 1000 ul tips (80 ul/s) and want to leave as close as possible to the original sample volumes (50, 100, 150, 200 µL) for the next staining step, while making sure we do not disturb the RBC pellet.
Right now, after intense liquid class optimization, I’m getting approximately the following averages:
• 50 → 67 µL
• 100 → 119 µL
• 150 → 164 µL
• 200 → 207 µL
Any further change of the liquid class leads to loss of red blood cells.
A few things I’ve noticed:
• When I use the virtual labware library to aspirate on the side, cLLD seems less sensitive and some wells retain more volume, which gives inconsistent results.
• I’ve found that aspirating in the center gives the most consistent liquid detection.
I wanted to ask:
-
Do you have any suggestions to reduce the residual volume further without risking the pellet?
-
Do you think it would be possible to detect the liquid in the center first, then slide toward the side wall while aspirating?
-
Or do you have any other ideas that might improve this step further? Like removing the bulk volume with 1000 ul and then do another pass with the 50 ul? We are washing the cells 3 times, so additional pipetting steps would increase the total run time.
Any advice would be greatly appreciated. Thank you in advance!
In regards to detecting the liquid height in the center of the well then moving to the side of the well, this is possible. The tips will retract to traverse height between the detection and the actual aspiration step, but it will still give the desired results. The aspiration can be setup with zero volume, then followed by the steps found in these posts (Capacitive liquid level detection (LLD) without aspiration/dispense and Finding the liquid height after aspiration and passing it on - #2 by Pascal) to retrieve a fixed height to be used in the following aspiration.
Pipetting remaining volume with a second tip could work, but I am in agreement that this would add unnecessary time to the overall run. Based on the volumes you’re working with, this should not be required and could run into similar issues with inconsistent volumes.
When I’ve worked on supernatant removals like these, I used a different technique that focused on using fixed heights instead of cLLD so that I could precisely control my volumes. This requires determining the height of the liquid in a well at the desired volume, then aspirating in one of the following ways:
-
Set the aspirate to a fixed height (no liquid following) to the calculated height and remove the supernatant. In some instances, the fixed height will need to be adjusted slightly higher or lower than the calculated height depending on the properties of the liquid. There may also be some additional liquid class property adjustments to ensure the pellet is not disturbed, but this technique removes a lot of variability. This technique can still be used it your target volumes have well-to-well variability, as pipetting steps can use an array of fixed heights similar to an array of volumes.
-
If the tips can not be plunged into the liquid to a fixed height like in option 1, then the calculated height can be used to determine a “false bottom” of the well that sits higher than the actual well bottom. When using liquid following, the tip will only travel as far as the inside bottom of a well, or to a container definition’s Max Pipetting Depth. This allows the tip to travel from the surface of the liquid to only as far as the calculated height. To implement this, either adjust the Max Pipetting Depth of the plate’s container or use a virtual shift to adjust the plate’s Z position at runtime. Note that this technique would require using one channel at a time if your target volumes have well-to-well variability.
Let me know if you have any questions.
Thank you,
Dan